Article Type: Case Report
Refractory gout: A case report and literature review
Yasmin Jaber1*; Maryia Vladykina1; Armenia Mordan2; David Podell, MD, PhD1
1Department of Internal Medicine, Yale School of Medicine, Waterbury, CT 06708, USA.
2Department of Internal Medicine, Danbury Hospital, Danbury, CT 06810, USA.
*Corresponding author: Yasmin Jaber
Department of Internal Medicine, Yale School of Medicine, 64 Robbins St, Waterbury, CT 06708, USA.
Received: Jan 22, 2025
Accepted: Feb 14, 2025
Published Online: Feb 21, 2025
Journal: Annals of Medical Case Reports and Reviews
Copyright: Jaber Y et al. © All rights are reserved
Citation: Jaber Y, Vladykina M, Mordan A, Podell D. Refractory gout: A case report and literature review. Ann Med Case Rep Rev. 2025; 1(1): 1004.
Abstract
Gout is characterized by monosodium urate crystal deposition, leading to painful inflammatory arthritis episodes, recurrent flares, tophi formation, and potential joint damage. This case study highlights a Samoan male experiencing persistent hyperuricemia despite standard treatments with allopurinol and colchicine. It reveals that traditional dosing strategies may be insufficient for certain ethnic groups, necessitating adjusted therapeutic approaches.
Furthermore, socio-economic factors can significantly complicate treatment adherence and exacerbate disease progression, underscoring the complex interplay between healthcare access and patient outcomes. In this instance, loss of medical insurance led to a worsening of symptoms due to medication non-adherence.
The case underscores the need for personalized treatment approaches that incorporate genetic, ethnic, and socio-economic considerations in managing refractory gout. In practice, general practitioners and rheumatologists should adjust treatment regimens based on ethnic-specific responses while integrating socio-economic support into medical care. Refining guidelines through research is essential to accommodate these diverse needs.
Introduction
Gout is currently the most common inflammatory arthritis worldwide, with an estimated prevalence of greater than 3% in the US, up to 13.9% in Māori, and 14.9% in Pacific Islanders, and a rising global burden of disease. Prevalence appears to be even higher in Māori and Pacific Island men over the age of 65. Māori and Pacific Island people also tend to suffer from an earlier age of onset of disease, more severe presentation of gout, accelerated rates of joint damage, and higher concentrations of Serum Urate (SU) and C-Reactive Protein (CRP) [1]. These factors combined lead to a higher pain burden, risk of disability, and treatment costs in this population [2].
Gout is a disease of Monosodium Urate (MSU) crystal deposition, which manifests as recurrent episodes of acute, painful inflammatory arthritis (flares), usually involving joints of the lower limbs. Patients are asymptomatic between flares, but likelihood of recurrence is higher with more severe hyperuricemia. Persistent hyperuricemia can lead to the development of tophi, chronic joint inflammation, and structural damage. Tophaceous gout is a late presentation of long-standing disease and poor serum urate control. Refractory gout is defined by progressive clinical manifestations of gout in the setting of persistent hyperuricemia, despite appropriate therapy [3].
Several demographic and environmental factors have been associated with hyperuricemia and gout, including ethnicity, older age, male gender, diet, smoking, alcohol consumption, and underlying comorbidities (i.e., hypertension, obesity, renal failure). While some may be independent risk factor for developing gout, they can also limit therapy (e.g., nonsteroidal antiinflammatory drugs or colchicine in patients with renal failure) [4,5].
An essential component of gout management is adequate control of SU levels. Several studies have shown that patients of Māori and Pacific Island descent may require higher dosing of allopurinol to achieve target concentrations [6]. We herein present the case of a Samoan male with tophaceous gout, refractory to standard medical therapy.
Case presentation
A 40-year-old male of Samoan ethnicity with a past medical history of hypertension, osteoarthritis of the knees, and very deforming tophaceous gout presented to our Rheumatology clinic for management of his gout after relocating from a different state. Relevant medical history included depression and obstructive sleep apnea (OSA). He had undergone gastric bypass surgery at the age of 20 and uvulopalatopharyngoplasty at the age of 17.
At the time of presentation, the physical exam revealed an obese male with Body Mass Index (BMI) of 45 kg/m2. Scattered tophi were noted over bilateral Metacarpophalangeal joints (MCPs) and Proximal Interphalangeal joints (PIPs); synovitis of right third PIP; reduced range of motion of 3rd and 5th PIP bilaterally, swelling of 5th digits consistent with dactylitis, and contracture of right 4th PIP. Reduced grip strength symmetrically bilaterally, markedly reduced range of motion of wrists bilaterally, with mild synovitis of the left wrist were noted as well. The elbows had scattered tophi bilaterally. At this time, he had been receiving allopurinol 100 mg daily and colchicine, with only slight improvement.
The patient was first diagnosed with tophaceous gout at the age of 17. He was treated with allopurinol but was taking it intermittently for the first several years. In later years, developed a particular rash on his left foot and, considering his joint involvement, was diagnosed with psoriatic arthritis. No skin biopsy was performed, but he was treated with methotrexate for about 4 months, with some improvement. Symptoms continued to be uncontrolled, and adalimumab was added. He noted improvement in the rash but not the joint pain. Shortly thereafter, he stopped taking the methotrexate but continued taking adalimumab. Despite reporting overall improvement, he was still requiring 20 mg of prednisone daily due to joint pain and stiffness, which had caused significant weight gain and hypertension. Although the diagnosis of gout was not proven by direct visualization of MSU crystals, the patient did have scattered tophi over his hands, elbows, ankles, stomach, and ears.
Due to losing medical insurance, the patient was unable to take any of his medication for some time, during which his condition further deteriorated, with a significant increase in joint pain and stiffness. When he regained medical insurance, he was again treated with adalimumab. He was restarted at a dose of 10 mg weekly, along with allopurinol 400 mg daily and colchicine 0.6 mg twice a day. In addition, he was restarted on prednisone at a reduced dose of 10 mg daily, with the intent to taper down. Due to persistent symptoms, allopurinol was titrated upwards to 800 mg daily.
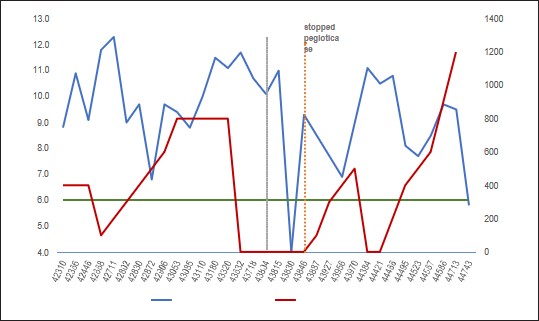
On high-dose allopurinol and colchicine therapy, the patient was reporting flares about once a year. He also continued to suffer from tophi formation, which was particularly bothersome. Plain film of the right foot was notable for extensive degenerative changes of the first and second metatarsophalangeal joints, and mild degenerative changes of the midfoot. Plain film of the hands revealed bilateral juxta-articular osteoporosis, osseous erosions at the 2nd PIPs, and 1st and 2nd carpometacarpal articulations. Given refractory erosive, tophaceous gout not responsive to high-dose allopurinol therapy, the patient was referred for pegloticase therapy. He stopped allopurinol months in advance while pending approval to start pegloticase, and in the interim, his symptoms progressed. He was having significant difficulty with Activities of Daily Living (ADLs) and had started using a wheelchair to mobilize.
The patient received three infusions of pegloticase, but it was discontinued due to rising concentrations of SU and a concern for risk of anaphylaxis. He was restarted on allopurinol 300mg daily and gradually increased to 900 mg daily after confirming HLAB 58:01 negative status.
At this point, the patient was tolerating allopurinol well with no adverse effects, and having great improvement in his mobility, despite noting new tophi formation. SU was still elevated at 9.7 mg/dL. The patient was then started on 1200 mg/dL of allopurinol daily, in addition to 500 mg of probenecid daily and colchicine 0.6 mg twice a day. This regimen, combined with increasing fluid intake, finally led to an SU level that was below target, at 5.8 mg/dL. Clinically he still suffered from new tophi formation. It was decided to continue the patient on this regimen along with close clinical monitoring.
Discussion
Several modifiable and nonmodifiable risk factors contributed to the challenges faced in our patient’s care. Below we will elaborate these risk factors and the role of each in hyperuricemia and gout.
Ethnicity, gender, and genetic risk
While incidence is progressively rising worldwide, certain populations have distinctly higher prevalence of hyperuricemia and gout [2]. This seems to be highest in Oceanic countries, specifically in indigenous and South Pacific Island populations [7]. In one study using the New Zealand national health database, all-ages prevalence of gout was found to be 2.69%, while prevalence of gout in the Māori and Pacific populations was over three-fold higher than those of European ancestry [1]. Prevalence was also notably higher in men, at 3.6 times more than women, and in the elderly. Worldwide, males seem to be affected between 2 and 11 times more than females, depending on the population studied [8-10].
Regarding genetic predisposition, previous studies have shown an association between genetic polymorphisms, urate concentrations, and risk of gout. In the Pacific Island population, two genes encoding urate transporters have been found to be associated with SU levels. They are the ABCG2 (minor allele rs2231242) and SLC2A9 genes. While SLC2A9 has been shown to be a risk factor for gout in both the Māori and Pacific Island people, ABCG2 rs2231142 has been associated with risk of gout in people of western (I.e., Tonga and Samoa) but not eastern (I.e., Māori, Cook Island) Polynesian ancestry [11,12].
ABCG2 encodes a urate transporter in the proximal renal tubule and may also mediate urate expression in the gut, as it is expressed in the liver and intestine. A study by Phipps-Green et al. found that at the minor allele rs2231242, Pacific Islanders had a high frequency of homozygosity, when compared to Māori and Cook Islanders.
SLC2A9 encodes Glucose Transporter type 9 (GLUT9) and has been shown to be a urate transporter, inhibitable by uricosuric agents such as probenecid and losartan [13,14]. In certain populations, an association has been proven between genomic variants of SLC2A9 and low fractional excretion of SU, which leads to hyperuricemia and gout [15]. In Pacific Island populations, SLC2A9 has been linked to gout susceptibility as well, though it may not be the sole etiologic agent [16].
Socioeconomic background
While historically gout has been associated with afluence and indulgence, recent studies have shown increased prevalence in areas of lower income and in people of lower socioeconomic status. There is also a higher prevalence in those with nonmanual skilled occupations compared to professional occupation [17]. Other studies suggest a lower risk of gout in rural residents than in urban resident [18]. Additionally, lower income may be associated with food insecurity or poor nutritional status and may portend barriers to access to healthcare and medication [19].
OSA
There are several pathophysiologic theories behind the association of gout and OSA [20]. It is thought to be related to hypoxia-induced increase in purine concentrations by increasing degradation of Adenosine Triphosphate (ATP), alternation of pH, leading to further crystal formation, and increases in uric acid renal absorption by increasing lactic acid production.
Risk factors in gout population frequently overlap between OSA and gout, making it hard to detect the true correlation. When matched to other risk factors, such as BMI, smoking status, history of heart failure etc., most of them suggest that correlation is true after matching [21,22]. Given multiple other gout-related factors in our patient, a confounding effect might be present.
Bariatric surgery
Interestingly, although some studies suggest that the early post-operational period after bariatric surgery might be associated with increased risk [23] of gout attacks, it was noted that in the long-term (>3 months postoperative), history of bariatric surgery was associated with decreased urate levels and gout attacks [24,25]. The observed effect is related to post-bariatric surgery weight loss and does not, reflect data on individuals with failed bariatric surgery, such as our patient. This may explain his severe course as well.
Guideline limitations
In the most recent guidelines issued by the American College of Rheumatology for the management of gout [26], treatment with allopurinol is recommended as the preferred first-line agent. The guidelines also recommend a treat-to-target strategy, defined as dose titration guided by SU levels, with a goal of <6 mg/dL. Allopurinol is very effective at decreasing SU levels, but the maximum FDA-approved dose is 800 mg/day [27]. As mentioned above, the patient in our case continued to suffer gout flares and develop new tophi, despite the high dose of allopurinol, even in combination with colchicine.
The recombinant form of uricase, pegloticase, is an alternative therapy developed for the treatment of individuals with chronic gout refractory to other urate-lowering agents [28]. While pegloticase treatment can lead to rapid clinical improvement and sustained reductions in SU, it is not without adverse effects. The most serious adverse effects of pegloticase therapy are immunogenicity, characterized by development of antibodies, and infusion reactions. A rise in SU indicates antibody development and therefore treatment failure and warrants discontinuation of treatment. Concomitant therapy with immunosuppressive agents to mitigate the immune response has been suggested, but further research is needed in this field [29].
In the case of our patient, his SU levels continued rising after starting pegloticase. Due to this and a concern for anaphylaxis, therapy was stopped, and he was restarted on allopurinol. The dose was again gradually increased with close monitoring of his symptoms and SU levels. After expert consultation, he was placed on the above-mentioned regimen, which included doses of allopurinol well-above the maximum approved dose. However, with this regimen, our patient did not suffer any adverse events and the SU levels were below 6 for the first time. We believe this to be the first documented case of successful treatment of refractory tophaceous gout with this regimen.
Conclusion
We have herein presented a challenging case of refractory tophaceous gout in a patient with several significant modifiable and nonmodifiable risk factors. Successful treatment was ultimately achieved by supratherapeutic dosing of allopurinol, along with other urate-lowering agents and close monitoring.
Management of gout requires a multi-pronged approach aimed at patients’ various risk factors. In the case of our patients, his Samoan ancestry, gender, and socioeconomic status leading to loss of insurance coverage were the major nonmodifiable risk factors. Alternatively, obesity and OSA are modifiable risk factors which may have contributed to his refractory gout. While most cases of gout are managed in the primary care setting, early rheumatology referral can help optimize therapy and prevent development of complications. This is especially important in those who develop gout at a young age, like our patient, as they will likely have to live with the consequences of gout for decades to come.
References
- Winnard D, Wright C, Taylor WJ, Jackson G, Te Karu L, Gow PJ, et al. National prevalence of gout derived from administrative health data in Aotearoa New Zealand. Rheumatology. 2012; 51: 901–9.
- Roddy E, Choi HK. Epidemiology of Gout. Rheumatic Disease Clinics of North America. 2014; 40: 155–75.
- Dalbeth N, Haskard DO. Mechanisms of inflammation in gout. Rheumatology. 2005; 44: 1090–6.
- Rothenbacher D, Primatesta P, Ferreira A, Cea-Soriano L, Rodriguez LAG. Frequency and risk factors of gout flares in a large population-based cohort of incident gout. Rheumatology. 2011; 50: 973–81.
- Dalbeth N, Kumar S, Stamp L, Gow P. Dose adjustment of allopurinol according to creatinine clearance does not provide adequate control of hyperuricemia in patients with gout. The Journal of rheumatology. 2006; 33: 1646–50.
- Dalbeth N, House ME, Horne A, Te Karu L, Petrie KJ, McQueen FM, et al. The experience and impact of gout in Māori and Pacific people: a prospective observational study. Clinical Rheumatology. 2012; 32: 247–51.
- Lennane GAQ, Rose BS, Isdale IC. Gout in the Maori. Annals of the Rheumatic Diseases. 1960; 19: 120–5.
- Kim JW, Kwak SG, Lee H, Kim SK, Choe JY, Park SH. Prevalence and incidence of gout in Korea: data from the national health claims database 2007–2015. Rheumatology International. 2017; 37: 1499–506.
- Kuo CF, Grainge MJ, Mallen C, Zhang W, Doherty M. Rising burden of gout in the UK but continuing suboptimal management: a nationwide population study. Annals of the Rheumatic Diseases. 2014; 74: 661–7.
- Kuo CF, Grainge MJ, See LC, Yu KH, Luo SF, Zhang W, et al. Epidemiology and management of gout in Taiwan: a nationwide population study. Arthritis Research & Therapy. 2015; 17: 13.
- B Phipps-Green AJ, Hollis-Moffatt JE, Dalbeth N, Merriman ME, Topless R, Gow PJ, et al. A strong role for the ABCG2 gene in susceptibility to gout in New Zealand Pacific Island and Caucasian, but not Māori, case and control sample sets. Human Molecular Genetics. 2010; 19: 4813–9.
- Roberts RL, Wallace MC, Wright DFB, Cadzow M, Dalbeth N, Jones PB, et al. Frequency of CYP2C9 polymorphisms in polynesian people and potential relevance to management of gout with benzbromarone. Joint Bone Spine. 2014; 81: 160–3.
- Hollis-Moffatt JE, Xu X, Dalbeth N, Merriman ME, Topless R, Waddell C, et al. Role of the urate transporterSLC2A9gene in susceptibility to gout in New Zealand Māori, Pacific Island, and Caucasian case-control sample sets. Arthritis & Rheumatism. 2009; 60: 3485–92.
- Caulfield MJ, Munroe PB, O’Neill D, Witkowska K, Charchar FJ, Doblado M, et al. SLC2A9 Is a High-Capacity Urate Transporter in Humans. Hattersley A, editor. PLoS Medicine. 2008; 5: e197.
- Hollis-Moffatt JE, Xu X, Dalbeth N, Merriman ME, Topless R, Waddell C, et al. Role of the urate transporterSLC2A9gene in susceptibility to gout in New Zealand Māori, Pacific Island, and Caucasian case-control sample sets. Arthritis & Rheumatism. 2009; 60: 3485–92.
- Dehlin M, Jacobsson L, Roddy E. Global epidemiology of gout: prevalence, incidence, treatment patterns and risk factors. Nature Reviews Rheumatology. 2020; 16: 380–90.
- Popert AJ, Hewitt J. Gout and Hyperuricaemia in Rural and Urban Populations. Annals of the Rheumatic Diseases. 1962; 21: 154–63.
- McCormick N, Lu N, Yokose C, Joshi AD, Sheehy S, Rosenberg L, et al. Racial and Sex Disparities in Gout Prevalence Among US Adults. JAMA Network Open. 2022; 5: e2226804.
- Khandwala P, Desai D, Sen M. Obstructive Sleep Apnea: A Contributing Factor in Gout. Cureus. 2024.
- van Durme C, Spaetgens B, Driessen J, Nielen J, Sastry M, Boonen A, et al. Obstructive sleep apnea and the risk of gout: a population-based case-control study. Arthritis Research & Therapy. 2020; 22.
- Khandwala P, Desai D, Sen M. Obstructive Sleep Apnea: A Contributing Factor in Gout. Cureus. 2024.
- Héctor Romero-Talamás, Daigle CR, Aminian A, Ricard Corcelles, Brethauer SA, Schauer PR. The effect of bariatric surgery on gout: a comparative study. Surgery for Obesity and Related Diseases. 2014; 10: 1161–5.
- Lu J, Bai Z, Chen Y, Li Y, Tang M, Wang N, et al. Effects of bariatric surgery on serum uric acid in people with obesity with or without hyperuricaemia and gout: a retrospective analysis. Rheumatology. 2021.
- Yeo C, Kaushal S, Lim B, Syn N, Oo AM, Rao J, et al. Impact of bariatric surgery on serum uric acid levels and the incidence of gout—A meta-analysis. Obesity Reviews. 2019; 20: 1759–70.
- FitzGerald JD, Dalbeth N, Mikuls T, Brignardello-Petersen R, Guyatt G, Abeles AM, et al. 2020 American College of Rheumatology Guideline for the Management of Gout. Arthritis Care & Research. 2020; 72: 744–60.
- Zyloprim (allopurinol) product information. East Brunswick (NJ): Casper Pharma; 2018. URL: https://www.accessdata.fda.gov/ drugsatfda_docs/label/2018/016084s044lbl.pdf.
- Schlesinger N, Lipsky PE. Pegloticase treatment of chronic refractory gout: Update on efficacy and safety. Seminars in Arthritis and Rheumatism. 2020; 50: S31–8.
- Botson JK, Tesser JRP, Bennett R, Kenney HM, Peloso PM, Obermeyer K, et al. Pegloticase in combination with methotrexate in patients with uncontrolled gout: A multicenter, open-label study (MIRROR). The Journal of Rheumatology. 2020; jrheum.200460.