Article Type: Case Report
Chimeric scapular free flap reconstruction for concurrent ameloblastoma and squamous cell carcinoma of the midface: A case report
Charles Witmer1; Michael Chang1; Russell Arellanes, DO2; Changxing Liu, MD, PhD3*
1Department of Biology, University of Pennsylvania, 433 S University Ave, USA.
2Department of Otolaryngology and Head and Neck surgery, Oklahoma State University, USA.
3Department of Otolaryngology – Head and Neck Surgery, Ascension St. John Medical Center, USA.
*Corresponding author: Changxing Liu
Department of Otolaryngology – Head and Neck Surgery, Ascension St. John Medical Center, USA.
Email ID: Changxing.Liu@Ascension.org
Tel: 918-403-6284
Received: Aug 22, 2025
Accepted: Sep 22, 2025
Published Online: Sep 29, 2025
Journal: Annals of Medical Case Reports and Reviews
Copyright: Liu C et al. © All rights are reserved
Citation: Witmer C, Chang M, Arellanes R, Liu C. Chimeric scapular free flap reconstruction for concurrent ameloblastoma and squamous cell carcinoma of the midface: A case report. Ann Med Case Rep Rev. 2025; 1(2): 1006.
Abstract
Ameloblastomas are benign neoplasms originating from various types of odontogenic epithelium, where squamous cell carcinomas are one of the most common malignant neoplasms of the oral cavity. The simultaneous occurrence of ameloblastomas with squamous cell carcinomas is rarely described in the literature. We present a case of a 52-year-old male presenting with locally destructive ameloblastoma and squamous cell carcinoma of the midface. Reconstruction was performed with a chimeric scapular free flap. Addressing these lesions requires comprehensive imaging interpretation and physical examination for early identification. As well as thorough preoperative planning and counseling for management of these lesions. We also discuss the utilization and versatility of chimeric scapular free flaps to reconstruct mid-face defects.
Keywords: Scapular free flap; Ameloblastoma; Squamous cell carcinoma; Oral cavity; Maxillary reconstruction; Chimeric free flap.
Introduction
Ameloblastomas are benign neoplasms originating from various types of odontogenic epithelium, including the epithelium from enamel, odontogenic cysts, and stratified squamous epithelium [1]. They represent 1% of oral tumors and 9-11% of odontogenic tumors; they are thought to grow slowly but be locally destructive [1,3]. Ameloblastomas most commonly occur in the mandible along the third molar region 80% of the time, with the remaining 20% occurring in the upper jaw [1]. They can increase in size to cause facial asymmetry, occlusion issues, and pathological fractures but are often asymptomatic, showing no evidence of swelling [1]. These odontogenic tumors frequently appear radiolucent on CT scans, making them difficult to distinguish them from cysts [2].
On the other hand, Squamous Cell Carcinoma (SCC) is the most common malignancy of the oral cavity. It is characterized by a high mortality rate, risk of recurrence, and metastasis [3]. There is a paucity of literature describing the simultaneous occurrence of these two neoplasms. Some literature proposes that squamous cell carcinoma-like neoplasms can arise secondary to malignant changes induced by ameloblastomas [4].
Today, we present a case of a patient who presented with extensive bony destruction of the midface secondary to concurrent ameloblastoma and SCC. The neoplasms eroded through the nasal bone, hard palate, and sinuses. This case report poses the question of whether the ameloblastoma may have coincided with the SCC or transformed into a SCC. While a direct cause of ameloblastoma is not known, many risk factors of SCC are known and could have contributed to this case. The defect, while extensive, required reconstruction with a chimeric scapular tip-free with a combined parascapular myocutaneous free flap.
Case presentation
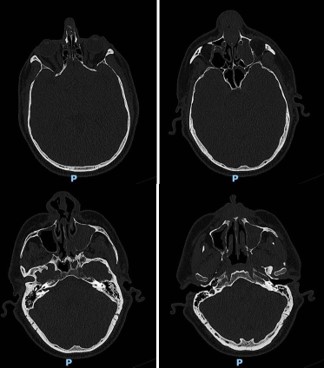
A 52-year-old male initially presented to the clinic for an evaluation of his nose. He had been suffering from nasal obstruction, rhinorrhea, anosmia, epistaxis, and chronic sinusitis for the last 5 years. At the time of presentation, he reported a change in the external aspect of his nose. He was unable to undergo endoscopic evaluation at the time, secondary to pain. STAT CT sinus with stealth protocol was performed. This initial CT scan revealed an opacified left maxillary sinus, polyposis vs thickening, and a small amount of tissue in the left nasal cavity.
The ostiomeatal complexes were occluded bilaterally, and there was bowing of the nasal septum to the right (Figure 1). He was scheduled for an endoscopic evaluation under anesthesia with nasal biopsy and frozen section analysis. Intraoperatively patient was noted to have fungating masses within bilateral nasal cavities. Multiple biopsies were obtained showing invasive squamous cell carcinoma. The case was terminated and a CT neck with contrast was obtained for surgical planning.
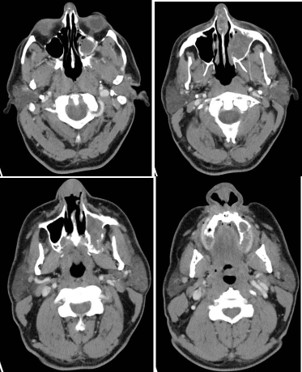
The new CT showed an ulcerated mass within the nasal cavity and a 1.4 cm septal perforation. Fluid and soft tissue were completely opacifying the left maxillary sinus. There was destruction of the nasal bone with the involvement of nasal skin, septal destruction with extension to the hard palate, and an extensive mass inside of the left maxillary sinus without any apparent orbital floor invasion (Figure 2). No cervical or distant metastases were appreciated at the time. The clinical Tumor, Nodal, and Metastasis (TNM) staging for the tumor was noted to be cT4N0M0. We recommended a left maxillectomy, left modified neck dissection with subtotal rhinectomy, and left chimeric scapular free flap for reconstruction.
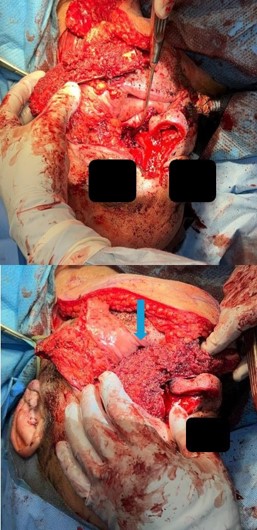
The procedure began with a left Weber-Ferguson incision. The visible abnormal skin was marked out and removed. This involved 70% of the nasal dorsum extending from the rhinion to the cephalic aspect of the lower lateral cartilages and nasal ala. An intraoral left gingival labial sulcus incision was made extending to the midline of the soft palate and laterally to the gingivobuccal sulcus. The periosteum was elevated with a periosteal elevator, and a reciprocating saw was used to make bony cuts. These were performed through the right lateral nasal bone, junction part between the right maxilla and right nasal bone, and extending into the lower piriform aperture. Another bony cut was made horizontally between the nasal bone and frontal bone superiorly. This was extended to the left medial canthus to include the left orbital medial orbital wall rim and left orbital floor. Lastly, the saw was used along the left anterior maxilla with the intention of removing the maxillary sinus wall completely. Intraoperatively, the tumor was noted to extend into the left parapharyngeal space. Dissection was carried out, staying outside the palpable primary tumor while leaving a sleeve of healthy margin. The bony cuts were then used to cut through the hard palate on the left side and posteriorly separated from the soft palate. No bone cuts were made along the left nasal wall to ensure an en bloc resection. Soft tissue structures were then dissected freely. With the nasal cavity exposed, the saw was used to cut through the hole in the nasal septum. There were multiple defects present, including a skin defect around 6×4 cm, a mucosal defect of 3×4 cm, a left midface defect of 8×6 cm, and a hard palate defect of 6×4 cm. Margins were then carefully obtained and showed no residual tumor. A left modified neck dissection was then performed, and vessels were located for microvascular anastomosis.
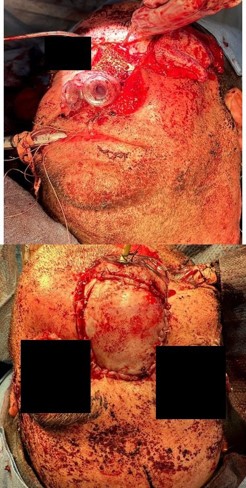
A chimeric scapular free flap was chosen for reconstruction because it provides multiple bony components and skin for reconstruction. A 10×2.5 cm segment of the lateral border of the scapula and entire scapular tip was harvested along with the subscapular muscle. The skin paddle obtained was parascapular. The two components of the flap were noted to have two separate arterial origins but shared venous drainage. The flap was then placed into the defect of the face. The bony aspect of the flap was utilized to reconstruct the hard palate and maxillary defect with osteotomies (Figure 3). Proximal scapular bone was plated to the residual hard palate and distal portion to the anterior maxillary process. Two nasal trumpets were placed through bilateral nasal vestibules. The middle portion of the skin paddle was de-epithelialized and used to pack into the nasal cavity. Medpor supported the left orbital contents and was secured to the scapular tip. The scapular tip was affixed to the left zygoma and frontal bone with a reconstruction plate. Then, titanium mesh was used for the nasal bony defect, and the skin paddle’s distal end covered it entirely (Figure 4). The microvascular anastomosis was then performed, drains and nasogastric tube were placed, and incisions were closed. The patient was admitted to the intensive care unit, where hourly flap perfusion checks were performed using a Cook arterial monitoring probe. The patient followed an uncomplicated post-operative course, with discharge on the seventh day.
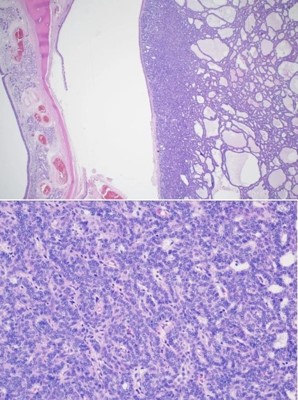
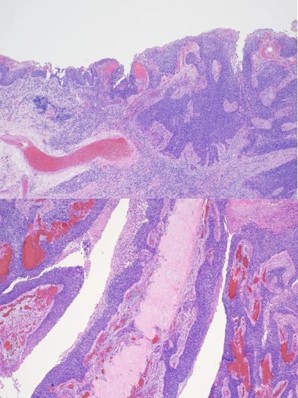
The patient was seen in an outpatient setting for a two week follow-up and discussion of final pathology results. These showed two masses: ameloblastoma with the invasion of the hard palate, posterior maxilla, and maxillary sinus (Figure 5) as well as a SCC with the cartilaginous and bony invasion of the nasal septum and nasal bone (Figure 6). During this clinic visit, he reported some discharge from one of the incisions and flap edema. He was otherwise doing well, with significant improvements in dysphagia and nasal obstruction. The flap adhered well to native tissue and appeared healthy. The infection cleared up over the next two weeks on a course of antibiotics, and the patient’s nasogastric tube was removed within the following three weeks.
The has been followed for 16 months and has continued to do well without evidence of recurrence. The patient is routinely followed with PET/CT scans. He has required a few procedures to revise his nasal dorsum but has otherwise had an unremarkable post-operative course. This case highlights the importance of careful imaging interpretation and physical examination for early identification and management of these lesions. It also highlights the utilization and versatility of chimeric scapular free flaps when reconstructing mid-face defects. Treatment algorithms for large mid-face defects vary widely from surgeon to surgeon, contingent on the surgeon’s expertise, training, and desired patient outcome. This case had a successful outcome using a chimeric scapular free flap, but other combinations of free flaps may pose favorable solutions for similarly complex defects.
Discussion/conclusion
We report a case of extensive nasal bone, hard palate, and sinus invasion from squamous cell carcinoma, which muddied the identification of an ameloblastoma. Simultaneous occurrences of ameloblastomas and SCC are rare, and it is difficult to appreciate both entities during initial examination. The ameloblastoma was present along the left maxillary sinus, posterior maxilla, and hard palate. Where moderately differentiated keratinizing squamous cell carcinoma was seen along the nasal bone and septal cartilage. In this case, metastatic disease was not found, and the margins were negative. The pathological staging was pT3N0M0 and pT4N0M0 for the ameloblastoma and squamous cell carcinoma, respectively. He was referred to radiation oncology and recommended to proceed with radiation therapy, but the patient opted not to pursue it. Previous case reports discuss the radiosensitivity of squamous cell carcinoma and the uncertainness of the role of radiation therapy in ameloblastomas [5,6].
Consent: An internal review board independently reviewed this study, and it did not require approval. Written informed consent for publication of this study was obtained from the patient.
References
- Masthan KM, Anitha N, Krupaa J, Manikkam S. Ameloblastoma. J Pharm Bioallied Sci. 2015; 7: S167-70.
- Mendenhall WM, Werning JW, Fernandes RD, Malyapa RS, Mendenhall NP. Ameloblastoma. Am J Clin Oncol. 2007; 30: 645-8.
- Imbesi Bellantoni M, Picciolo G, Pirrotta I, Irrera N, Vaccaro M, Vaccaro F, Squadrito F, Pallio G. Oral cavity squamous cell carcinoma: an update of the pharmacological treatment. Biomedicines. 2023; 11: 1112.
- Rais R, El-Mofty SK. Malignant transformation of a desmoplastic ameloblastoma to squamous cell carcinoma: a case report. Head Neck Pathol. 2019; 13: 705-10.
- Nishimura T, Nagakura R, Ikeda A, Kita S. Simultaneous occurrence of a squamous cell carcinoma and an ameloblastoma in the maxilla. J Oral Maxillofac Surg. 2000; 58: 1297–300.
- Matear DW, Crewe TC. Malignant ameloblastoma? A case report and review of literature outlining problems in diagnosis and treatment. J R Nav Med Serv. 1991; 77: 5-10.